



















Which species is the conjugate base of NH3. NH2-Correctly identify the correct conjugate acid-base pairs in the flowing equation: HC2H3O2(I) + H2O(I) ⇌ H30^3(aq) + C2H3O2-(aq) Quizlet Live. Quizlet Learn. Diagrams. Flashcards. Mobile. Help. Sign up. Help Center. Honor Code. Community Guidelines. Conjugate acids and bases are part of the Bronsted-Lowry theory of acids and bases. According to this theory, the species that donates a hydrogen cation or proton in a reaction is a conjugate acid, while the remaining portion or the one that accepts a proton or hydrogen is the conjugate base. The conjugate base may be recognized as an anion. NH3 + H2O <==> H3O+ + NH2-NH2- is therefore the conjugate base of ammonia. HPO4^2- is also amphoteric. It can donate a proton as an acid, or accept a proton to make behave as a base. To have a... Answer to 3.) Give the formula for the conjugate base of each species: (a) H2S; (b) HCN; (c) HSO4−. (a) (b) (c)... Start studying Conjugate Acid-Base Pairs/Lewis definition slides. Learn vocabulary, terms, and more with flashcards, games, and other study tools. (e) a species that can accept a pair of electrons. 2. In the Bronsted-Lowry system, a base is defined as: (a) a proton donor. (b) a hydroxide donor. (c) an electron-pair acceptor. (d) a water-former. (e) a proton acceptor. 3. In the equation: HF + H 2 O H 3 O + + F-(a) H 2 O is a base and HF is its conjugate acid. (b) H 2 O is an acid and HF is The amide ion, NH_2^-. The conjugate base of any species is that species less a proton. Ammonia less a proton is the amide ion, NH_2^-. Mass and charge are conserved as always. This species does NOT exist in water, but it is the characteristic anion in liquid ammonia, a water like solvent that will support more powerful bases than does water. Q1. conjugate base --> is the base which forms when NH3 acts as an acid so, for NH3 to act as an acid, it must lose 1 H+ NH3 --> NH2- + H+ then NH2- is the base choose C Q2 note view the full answer The Arrhenius definition of acid and base is limited to aqueous (that is, water) solutions. Although this is useful because water is a common solvent, it is limited to the relationship between the H + ion and the OH − ion. What would be useful is a more general definition that would be more applicable to other chemical reactions and, importantly, independent of H 2 O. The species remaining after a Brønsted-Lowry acid has lost a proton is the conjugate base of the acid. The species formed when a Brønsted-Lowry base gains a proton is the conjugate acid of the base. Thus, an acid-base reaction occurs when a proton is transferred from an acid to a base, with formation of the conjugate base of the reactant acid and formation of the conjugate acid of the reactant base.
[index] [2307] [9614] [3599] [8109] [2920] [8040] [9843] [2845] [5744] [3933]
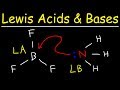
About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features Press Copyright Contact us Creators ... For example, OH− and NH3 are Lewis bases, because they can donate a lone pair of electrons. In the adduct, the Lewis acid and base share an electron pair furnished by the Lewis base. Usually the ... This organic chemistry video tutorial provides a basic introduction into lewis acids and bases. It explains how to predict the products of a lewis acid-base... Acid Base Equilibrium Organic Chemistry (Part 1) which side of an acid base reaction is favored HOW TO PREDICT THE EQUILIBRIUM DIRECT... high school chemistry http://leah4sci.com/aminoacids presents: Isoelectric Point of Amino Acids detailed tutorial with time-saving MCAT ShortcutIs your MCAT just around the corner... Introduction to conjugate acids and bases. Created by Sal Khan.Chemistry on Khan Academy: Did you know that everything is made out of chemicals? Chemistry is... Use Bronsted Lowry Acid/Base Theory to identify conjugate acid base pairs.More free chemistry help at www.chemistnate.com How can you figure out which chemical is the Lewis acid and Lewis base in a chemical reaction?Free chemistry help @ www.chemistnate.com To tell if NaCl (Sodium chloride) forms an acidic, basic (alkaline), or neutral solution we can use these three simple rules along with the neutralization re...
Copyright © 2024 top.tabking.online